
Written by Anela Ivanova, Department of Chemical and Pharmaceutical Engineering; Department of Physical Chemistry, Faculty of Chemistry and Pharmacy, Sofia University, Bulgaria
We recently discovered that drops of typical oils, such as those composed of alkane molecules, exhibit a spectacular series of shape transformations upon cooling, when stabilized with appropriate surfactants (Figure-1). The drops just need to be dispersed down to micrometer size. The shapes encompass regular polyhedra, platelets, and long fibers or drops bursting into nanoparticles.
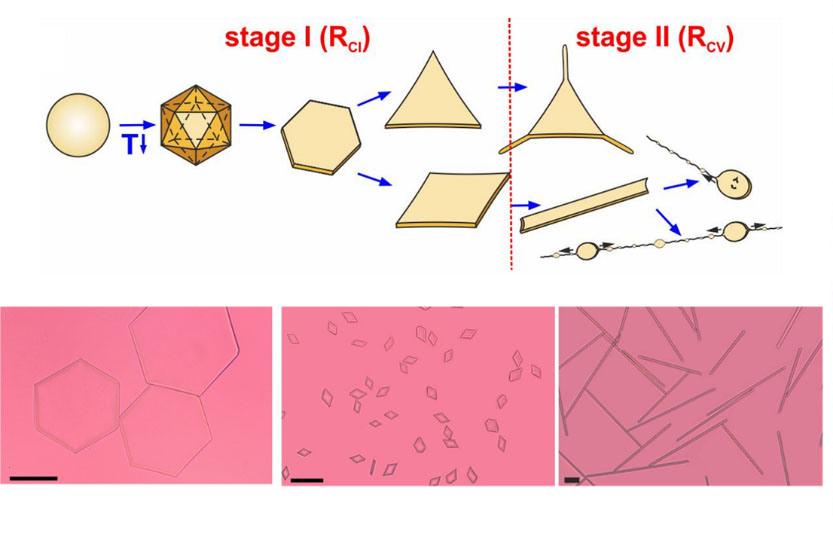
Figure 1: Schematic representation (top) and microscope images (bottom) of the experimentally observed shapes upon freezing of surfactant-stabilized alkane drops
Theoretical analysis suggested that the different shapes are caused by freezing layers of long-chain surfactants, adsorbed at the alkane-water interface, acting as a two-dimensional template for ordering of neighboring alkane molecules into thin surface multilayers. The drop dynamics showed that the alkane multilayers resembled “intermediate rotator phases”, i.e., semi-ordered structures where the molecules are aligned with respect to each other at long distances, like in crystals, but still preserve some disorder by rotating along their long axes. Revealing how alkane molecules act upon cooling might aid the design of different multicomponent surface-active formulations, the production of ‘clever’ nanoparticles, or facilitate the understanding of the interaction and absorption of some drugs in the living organisms. However, the rotator phases are in many cases short lived and difficult to register experimentally.
In order to check the templating effect of the surfactants and the presence of rotator phases during the very initial stages of freezing of alkanes, computational modelling was done on model oils comprised solely of hexadecane or of hexadecane stabilized by oligoethylene oxide surfactants (C16EO2) at the interface with water (Figure 2). Fully atomistic molecular dynamics (MD) was used to monitor the behaviour of the systems upon melting and freezing.
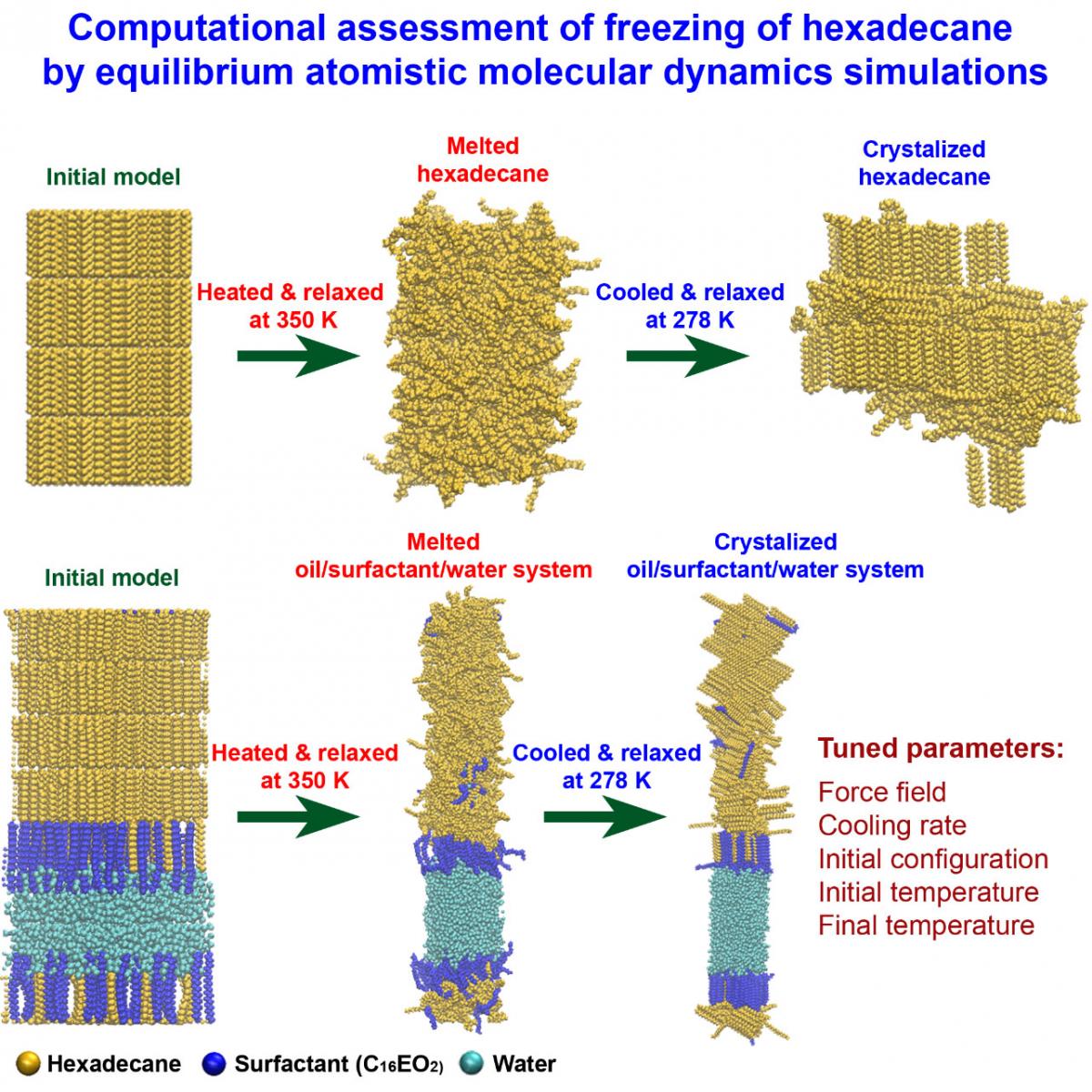
Figure 2: Illustration of the molecular dynamics procedure developed to study the initial stages of freezing of hexadecane-containing systems; representative structures in liquid and frozen state are shown
The resources provided by the ICEI/Fenix infrastructure were indispensable in this study. The time allocated at the Piz Daint supercomputer of the CSCS (Switzerland) within an ICEI project allowed running multiple MD simulations to tune a computational protocol, which is non-trivial for phase change studies. This included selection of an appropriate classical force field, determining the temperature of melting and freezing of the bulk and interface-separated systems, discerning the influence of the cooling rate and of the interface curvature (flat vs. circular) on the mechanism of crystallization and on the type and number of the obtained crystallites. A number of independent trajectories were also generated for each computational setup to verify statistically the outcome of the simulations.
The results obtained so far stand for a stochastic mechanism of freezing of bulk hexadecane, irrespective of the cooling rate. Crystallites nucleate at random locations in the bulk and start growing fast within tens of nanoseconds. They orient differently in space. The same is largely valid for surfactants-stabilized hexadecane at the interface with water. It is observed that indeed surfactant molecules freeze first already during the fast (< 1 ns) cooling stage of the simulations. This is then followed by stochastic freezing of hexadecane, the nuclei arising more or less stochastically in the vicinity of the surfactant molecules. Curving of the interface did not result in notable changes of the freezing mechanism or time scale. The presence of rotator phases is indicated by analysis of intermolecular radial distribution functions. More detailed and robust quantification to determine the types and persistence of these structural assemblies is underway.
Results stemming from the study included in the ICEI project were reported by the young members of the research team at the Bunsen-Tagung 2021 - Multi-Scale Modelling & Physical Chemistry of Colloids.
Acknowledgements:
This research has received funding from a VIHREN grant of the Bulgarian Ministry of Education and Science (Project ROTA-Active).
References:
- N. Denkov, S. Tcholakova, I. Lesov, D. Cholakova, S.K. Smoukov, Self-shaping of oil droplets via the formation of intermediate rotator phases upon cooling, Nature. 528 (2015) 392-395.
- D. Cholakova, N. Denkov, S. Tcholakova, I. Lesov, S.K. Smoukov, Control of drop shape transformations in cooled emulsions, Adv. Colloid Interface Sci. 235 (2016) 90-107.
- P. A. Haas, R. E. Goldstein, S. K. Smoukov, D. Cholakova, N. Denkov, Theory of shape-shifting droplets, Phys. Rev. Lett. 118 (2017) 088001.
- D. Cholakova, N. Denkov, Rotator Phases in Alkane Systems: In Bulk, Surface Layers and Micro/nano-confinements, Adv. Colloid Interface Sci. 269 (2019) 7-42.
- C. G. Wilson, L. Aarons, P. Augustijns, J. Brouwers, A. S. Darwich, T. De Waal, G. Garbacz, S. Hansmann, D. Hoc, A. Ivanova, M. Koziolek, Ch. Reppas, Ph. Schick, M. Vertzoni, J. A. García-Horsman, Integration of advanced methods and models to study drug absorption and related processes: An UNGAP perspective, Eur. J. Pharm. Sci. (2021) in press (DOI: 10.1016/j.ejps.2021.106100)
Participating labs: